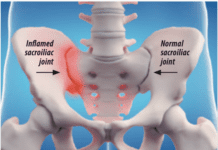
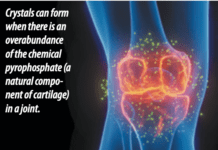
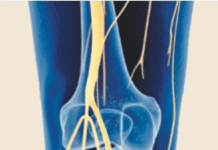
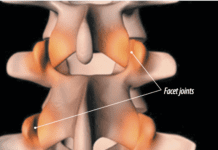
Add celecoxib (Celebrex) to the list of medications that may counter the effects of small, daily doses of aspirin used by millions of Americans to reduce the risk of heart disease and stroke. "There is evidence from laboratory testing that many arthritis drugs including naproxen (Naprosyn), ibuprofen (Motrin), and celecoxib (Celebrex) interfere with the blood-thinning effect of aspirin," says Steven E. Nissen, MD, Chairman, Department of Cardiovascular Medicine at Cleveland Clinic. "However, this has never been confirmed in prospective randomized trials." A study published in the January 2008 issue of the Journal of Clinical Pharmacology confirmed previous research that ibuprofen undermines aspirins ability to act as an anti-platelet agent.
To continue reading this article or issue you must be a paid subscriber.
Sign in